When Prof. Marc F. Schetelig takes visitors through his laboratories at the Justus-Liebig-University Gießen and the Fraunhofer Institute for Molecular Biology and Applied Ecology IME in Gießen, one of the things he shows them is a climate chamber that is kept at a temperature of 27 degrees Celsius. Inside are thousands of tiger mosquitoes (Aedes aegypti) in net cages. “We have to keep the females on a diet of cow’s or pig’s blood, served at 37 degrees Celsius, which we get directly from the abattoir,” says Schetelig. “This helps them lay their eggs, which we then put in bowls of water, where the larvae hatch and pupate.”
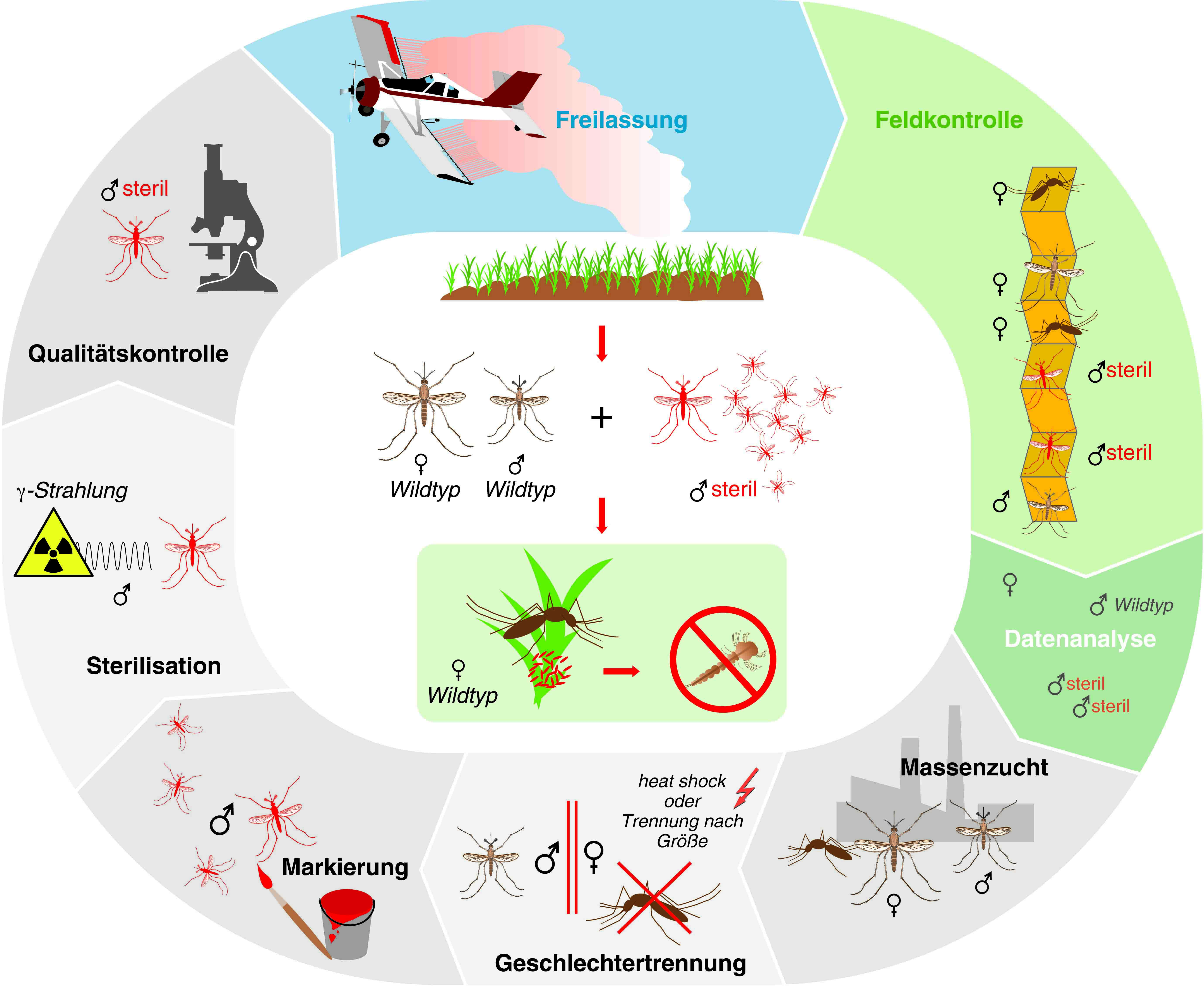
Schetelig is an expert on the sterile insect technique, or SIT, in which the mass release of sterile males decimates the population of insect pests (see box). With his team of researchers at the LOEWE Center for Insect Biotechnology & Bioresources, which is sponsored by the German state of Hesse, Schetelig is developing methods that increase the effectiveness of the sterile insect technique and allow it to be transferred to other insect pests.
A genetic on and off switch
Schetelig’s highest achievement to date has been his work on the spotted-wing drosophila vinegar fly, Drosophila suzukii, which comes from Southeast Asia and is spreading worldwide. The females lay their eggs in ripe fruit just before the harvest, when the farmers are generally no longer allowed to spray insecticide. This means that the fly can evade even efficient insecticides and at present, an effective control strategy is not on the market. In 2014, the spotted-wing drosophila caused massive crop failures to cherries, raspberries, plums and grapes in many parts of Central Europe. Mass propagation of the insects was feared for 2015, but was derailed by the hot, dry summer. This year, the wet early summer weather could once again provide the ideal conditions for a further spread.
Schetelig has designed a genetic system that causes the flies’ offspring to not survive beyond the embryonic stage. If male flies carrying this system mate with naturally occurring females, the fertilized eggs will not produce larvae. But the genetic system also includes a switch that can be used to turn the program off. This switch can be activated by feeding the flies the antibiotic tetracycline: the tetracycline containing diet allows the flies to be successfully rearing in the laboratory. “The new method doesn‘t require antibiotics for larval rearing and only a little amount of antibiotics is needed for the adult flies. So no antibiotics are transferred to the environment,” says Schetelig.
To help with mass breeding, the research team anchored yet another genetic system within the genome that can cause all females to die at the embryonic stage. “With this sexing system, we can considerably improve the effectiveness of mass breeding, because we raise only the males. Females need to be sorted out,” explains Schetelig. In the majority of conventional breeding methods, the females needed to be weeded out, sometimes manually, before a mass release.
Curbing dengue and Zika viruses
The team is currently working on transferring the genetic systems it developed for the spotted-wing drosophila to the tiger mosquito. It is hoped that this use of the sterile insect technique will help stop the tiger mosquito from continuing to spread a number of dangerous viruses. In addition to the dengue and Zika viruses, the tiger mosquito can also carry the viruses for yellow fever, Chikungunya and Rift Valley fever. Despite being indigenous to the tropics, this insect spread across the world. A related Aedes species has already established itself in Italy. And the first case of tiger mosquitoes surviving the German winter and hatching has been reported near Freiburg im Breisgau.
In addition to his high hopes in the new technology, Schetelig is also looking into the risks of releasing transgenic insects. As part of a project funded by the German Research Foundation DFG through the Emmy Noether Programme, he is examining for example how stable the integration of foreign genes into genomes really is and what happens when other animals eat the genetically modified insects.
“Since we use radioactive radiation to sterilize the males before their release, we are convinced that they will not multiply,” says Schetelig. He expects the technology will most likely find large-scale application in countries that already have regulations for the release of genetically modified organisms in place, such as in South America and to some extent in the United States. “The decisive factor in terms of acceptance is the level of threat posed by the pests,” Schetelig says. “If we had the same problems with dengue fever and Zika virus in Germany as they have in Brazil, we would also be searching for and evaluating different solutions. And the sterile insect technique with transgenic mosquitoes would definitely be part of the discussion.”
 Fraunhofer Institute for Molecular Biology and Applied Ecology IME
Fraunhofer Institute for Molecular Biology and Applied Ecology IME