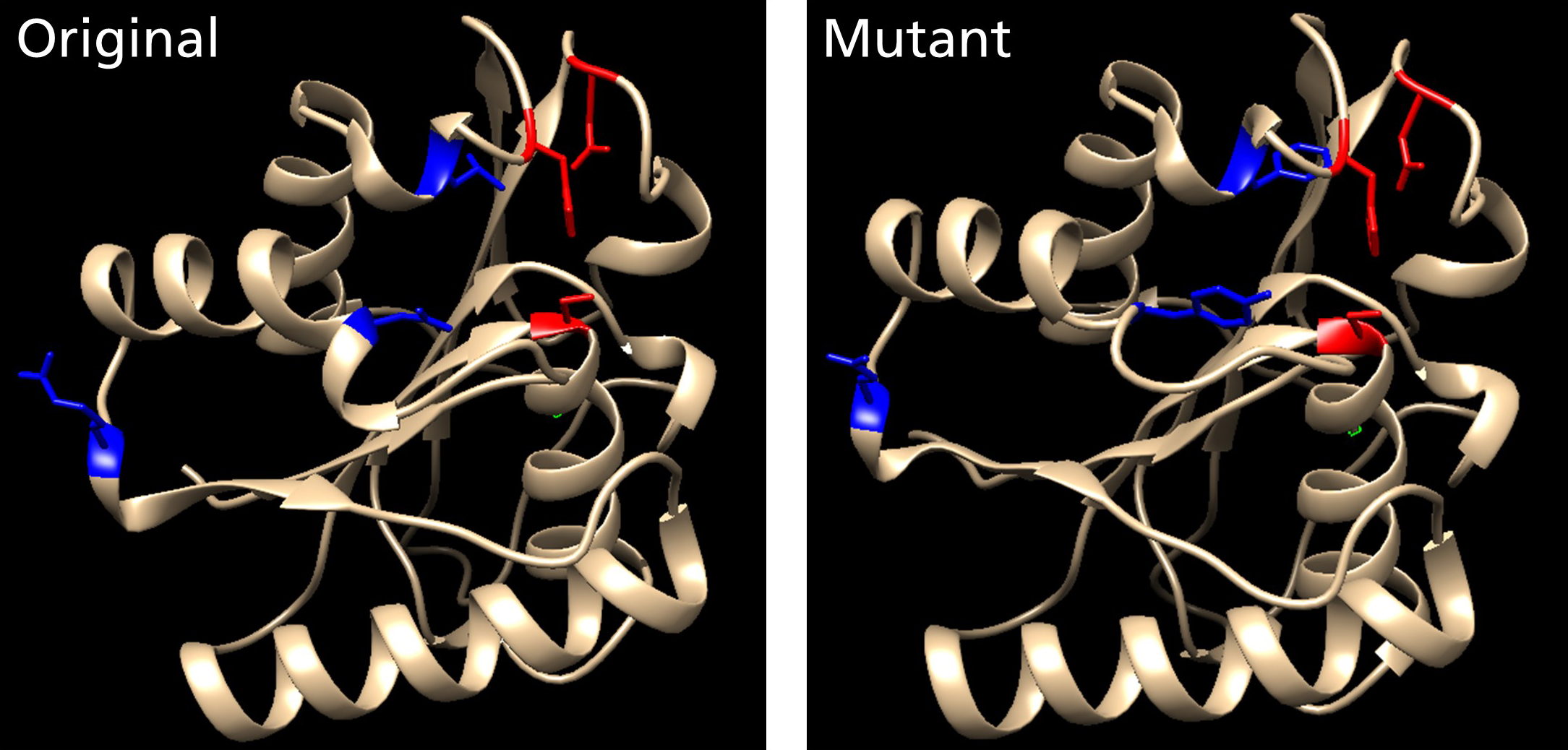
Enzymes catalyze specific chemical reactions under mild conditions and therefore have the potential to replace the chemical catalysts used in industry while offering further advantages such as greater specificity, lower toxicity and more environmental sustainability. However, natural enzymes are not always suitable for industrial processes and must be tailored for the required specifications. This can be achieved using protein-accelerated evolution, a process in which libraries of mutants are generated and screened for improved properties in iterative cycles.
The key element of directed evolution is the screening step because a suitable assay must be developed to highlight the property of interest that needs to be selected. We work with artificial compartments based on water-in-oil emulsions that act as pico-reaction vessels, allowing us to evaluate more than ten million enzyme variants in less than one hour. Each droplet contains one enzyme variant and the substrate needed for the synthesis of a fluorescent product using the catalytic activity of the enzyme. Droplets containing the most appropriate enzyme variants can be isolated by flow cytometry/florescence activated cell sorting or microfluidic devices.
We have developed successful screening systems based on flow cytometry and/or microfluidic devices for several enzymes classes, including glucose oxidases, cellulases, xylanases, chitinases, peroxidases and cellobiose dehydrogenases.
 Fraunhofer Institute for Molecular Biology and Applied Ecology IME
Fraunhofer Institute for Molecular Biology and Applied Ecology IME